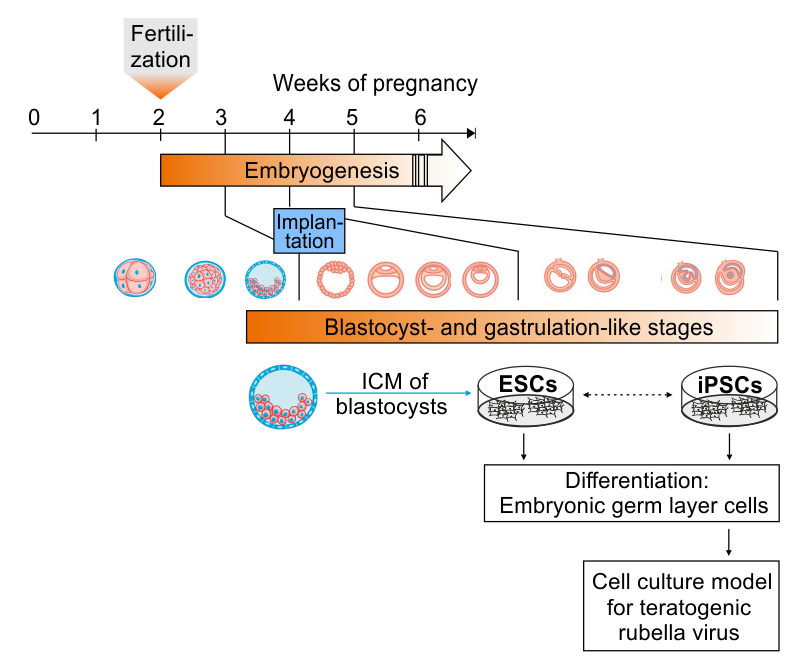
iPSC Platform
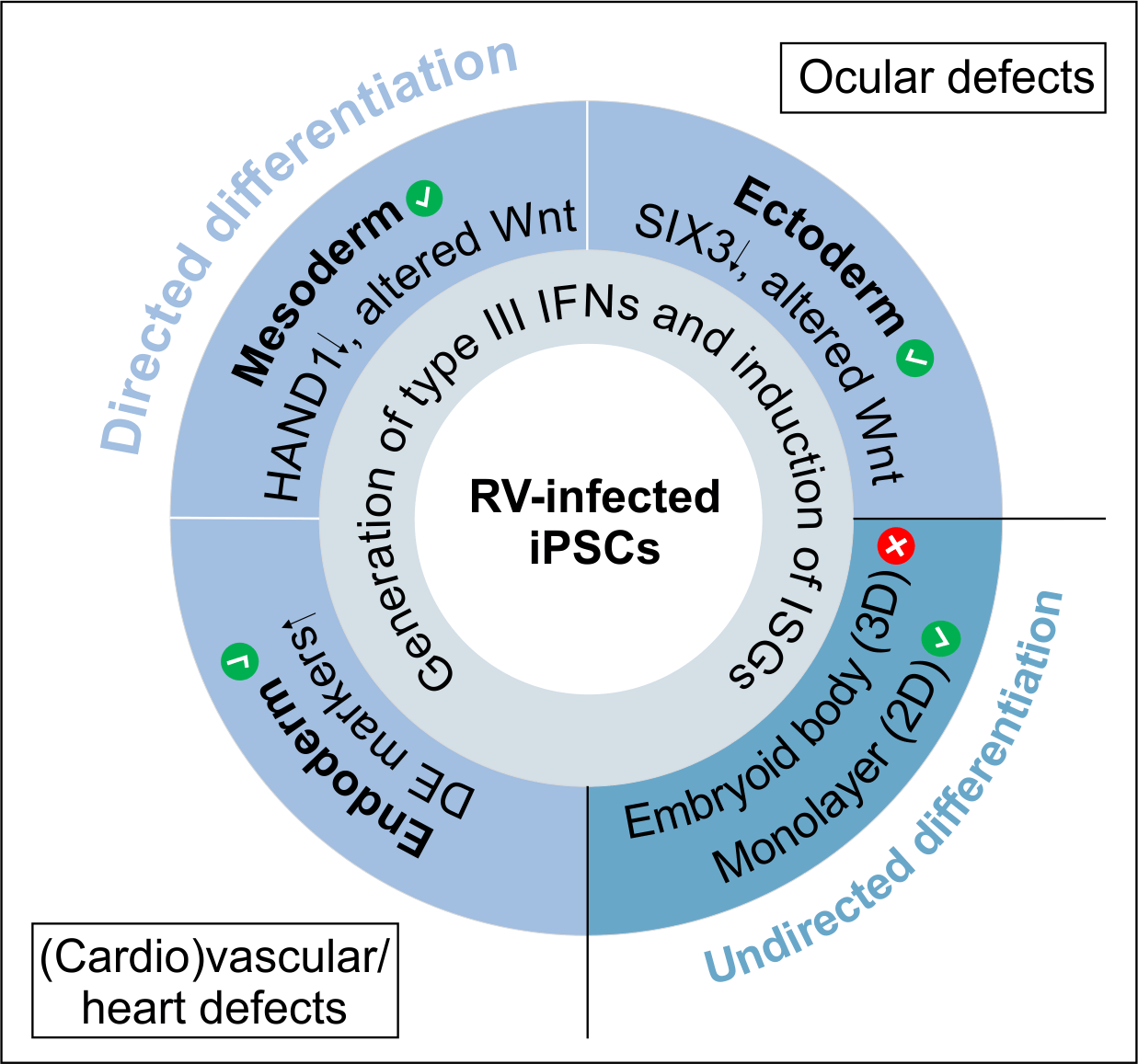
The ectoderm is involved in eye development, which is altered during congenital rubella virus infection. During ectodermal differentiation of rubella virus-infected cells eye field transcription factors such as SIX homeobox 3 (SIX3) and retina and anterior neural fold homeobox (RAX) are reduced in comparison to the uninfected population. During directed differentiation of RV-infected iPSCs the endoderm appeared to be the most affected cell population. The increased expression of markers for definitive endoderm such as Eomesodermin (EOMES) and Cerberus (CER) could affect its crosstalk with the ectoderm and especially the mesoderm during embryonal development. The mesoderm is involved in cardiac development, which is affected during congenital rubella virus infection. For more details please refer to our publication (Bilz et al., 2019).
Bilz NC*, Willscher E*, Binder H, Böhnke J, Stanifer ML, Hübner D, Boulant S, Liebert UG, Claus C. Teratogenic rubella virus alters the endodermal differentiation capacity of human induced pluripotent stem cells. Cells. 2019; 8(8). pii: E870. doi: 10.3390/cells8080870.
Claus C, Jung M, Hübschen JM. Pluripotent stem cell-based models: a peephole into virus infections during early pregnancy. Cells. 2020, 9, 542. doi: 10.3390/cells9030542.
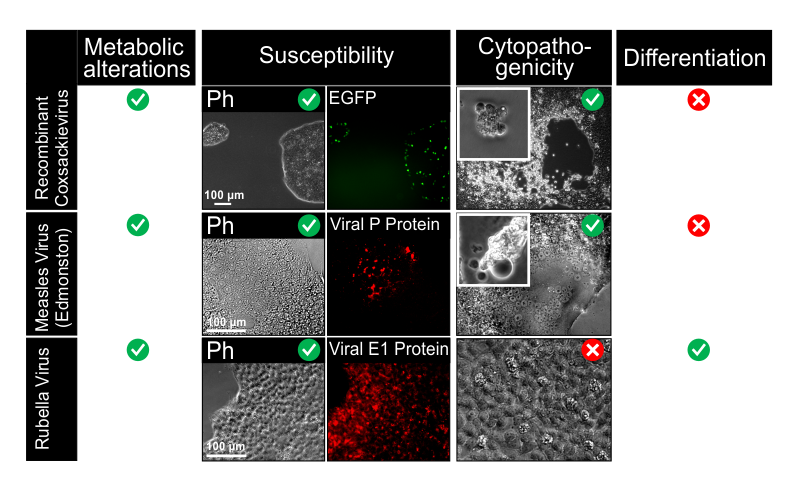
Hübner D, Jahn K, Pinkert S, Böhnke J, Jung M, Fechner H, Rujescu D, Liebert UG, Claus C. Infection of iPSC Lines with Miscarriage-Associated Coxsackievirus and Measles Virus and Teratogenic Rubella Virus as a Model for Viral Impairment of Early Human Embryogenesis. ACS Infect Dis. 2017 Dec 8;3(12):886-897. doi: 10.1021/acsinfecdis.7b00103. Epub 2017 Oct 26.
