Interaction with Mitochondria
Rubella virus (RV) is a rather slow replicating virus. This raised the hypothesis of a constant energy demand posed by RV on the respective host cell. Its low level of cytopathogenicity in turn could support the alteration of metabolic pathways by RV to ensure availability of metabolic substrates as building blocks for its progeny and the provision of the required energy (Claus & Liebert, Archives of Virology, 2018). Three approaches were pursued to highlight the importance of cellular metabolism, especially mitochondria-based pathways, for RV replication.
- The general importance of mitochondria for RV replication was shown through knock-down of the cellular protein p32 (also known as gC1q-R and C1QBP) by adenoviral vectors in Vero cells and the infection of a so called rho zero cell line (Claus et al., Journal of Virology, 2011). The protein p32 can be found in different cellular compartments, including mitochondria. The knock-down of p32 reduced RV replication and titer. Moreover, perinuclear cluster formation of mitochondria, which is usually noted for RV-infected Vero cells, was lost. The infection of the rho zero derivative of the 143B.TK- osteosarcoma cell line reduced RV titer by almost two log steps as compared to the parental cell line. Rho zero cells lack mitochondrial DNA and thus have no functional mitochondrial respiration.
- The activity of mitochondrial respiratory chain complexes I to IV was spectrophotometrically determined in RV-infected cells in comparison to the mock (uninfected) control (Claus et al., Journal of Virology, 2013). These assays revealed an increased activity of mitochondrial respiratory chain complex II during RV infection. This effect was noted for Vero, human adenocarcinoma cell line A549 and the human breast carcinoma cell line MCF-7 and thus occured in a cell line-independent manner.
- A bioeneregetic profile was generated for RV-infected Vero, A549 and human umbilical vein cells (HUVEC) as compared to the mock-infected controls by extracellular flux analysis using the Agilent Seahorse XFp Analyzer (Bilz & Jahn et al., Journal of Virology, 2018). Under both, basal (normal) and stressed conditions, RV induced a shift of its host cell to a higher energetic level. This involved an increase in glycolysis as well as in mitochondrial respiration. Figure 1 exemplarily shows the measurement cycle for mitochondrial respiratory function through the oxygen consumption rate (OCR) during basal and stressed conditions by extracellular flux analysis.
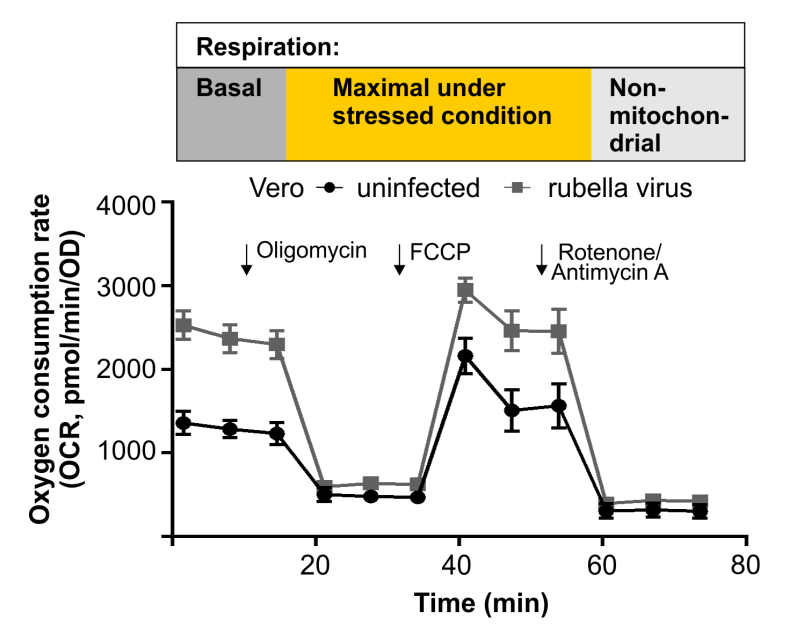
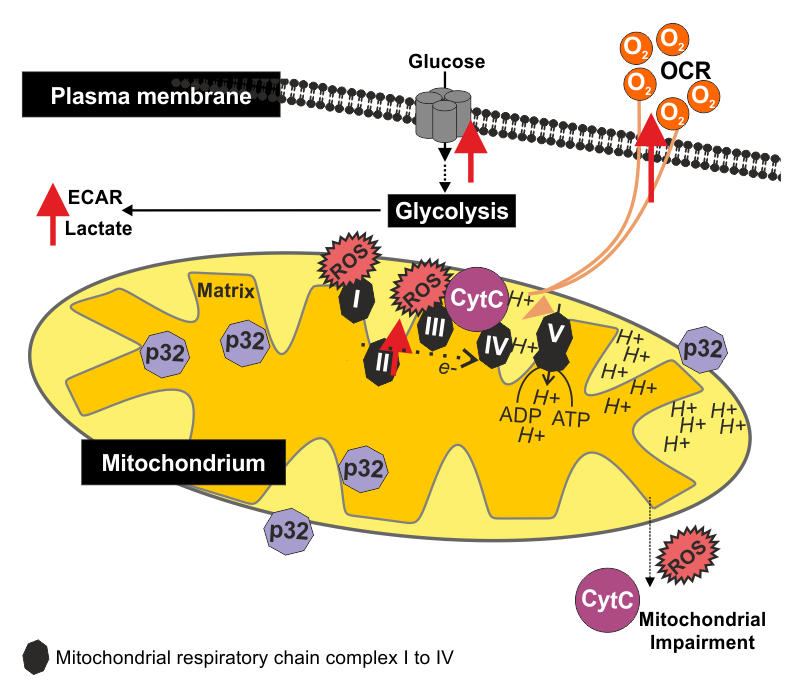
Figure 2 summarizes the findings of these three approaches, which have helped to elucidate the importance of mitochondrial functions for RV replication.
Bilz NC, Jahn K, Lorenz M, Lüdtke A, Hübschen JM, Geyer H, Mankertz A, Hübner D, Liebert UG, Claus C. Rubella Viruses Shift Cellular Bioenergetics to a More Oxidative and Glycolytic Phenotype with a Strain-Specific Requirement for Glutamine. J Virol. 2018; 92(17). pii: e00934-18. doi:10.1128/JVI.00934-18. 2018.
Claus C, Chey S, Heinrich S, Reins M, Richardt B, Pinkert S, Fechner H, Gaunitz F, Schäfer I, Seibel P, Liebert UG. Involvement of p32 and Microtubules in Alteration of Mitochondrial Functions by Rubella Virus. J Virol. 2011; 85(8):3881-92. doi: 10.1128/JVI.02492-10.
Claus C, Liebert UG. A Renewed Focus on the Interplay Between Viruses and Mitochondrial Metabolism. Arch Virol. 2014; 159(6):1267-77. doi:10.1007/s00705-013-1841-1.
Claus C, Schönefeld K, Hübner D, Chey S, Reibetanz U, Liebert UG. Activity Increase in Respiratory Chain Complexes by Rubella Virus with Marginal Induction of Oxidative Stress. J Virol. 2013; 87(15):8481-92. doi: 10.1128/JVI.00533-13.
